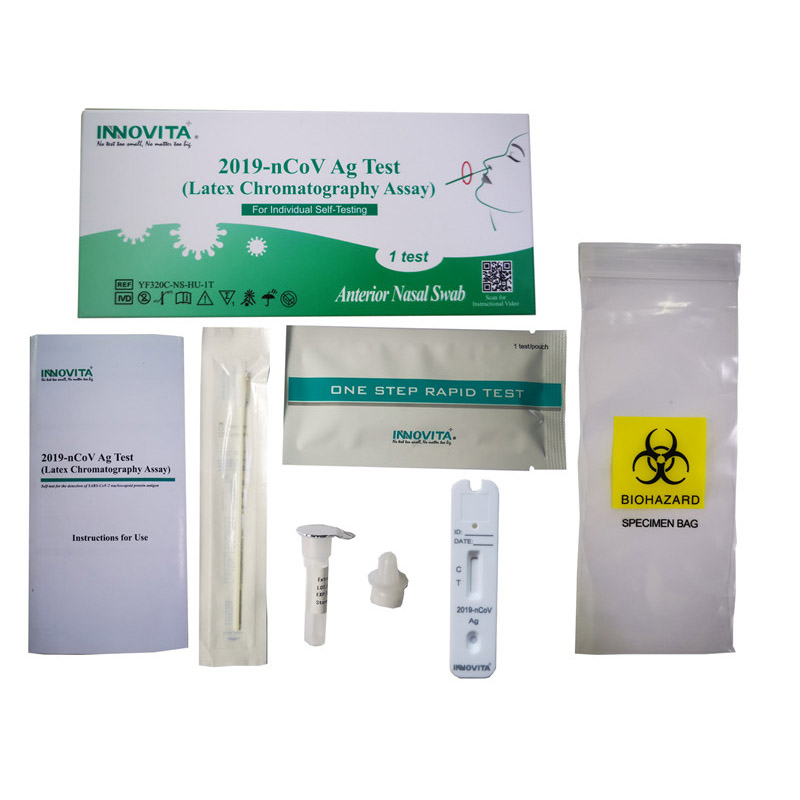
CE Certification Parainfluenza Virus Type 1 Manufacturers Exporters – 2019-nCoV Ag Test (Latex Chromatography Assay) / Professional Test / Anterior Nasal Swab – INNOVITA
CE Certification Parainfluenza Virus Type 1 Manufacturers Exporters – 2019-nCoV Ag Test (Latex Chromatography Assay) / Professional Test / Anterior Nasal Swab – INNOVITA Detail:
Product Detail:
Innovita® 2019-nCoV Ag Test is intended for the direct and qualitative detection of SARS-CoV-2 nucleocapsid protein antigen in anterior nasal swabs from individuals who are suspected of COVID-19 by their healthcare provider within the first seven days of the onset of symptoms or for screening of individuals without symptoms or other reasons to suspect COVID-19 infection.
The test results of this kit are for clinical reference only. It is recommended to conduct a comprehensive analysis of the condition based on the patient’s clinical manifestations and other laboratory tests.
Principle:
The kit is a double antibody sandwich immunoassay based test. The test device consists of the specimen zone and the test zone. The specimen zone contains monoclonal antibody against the SARS-CoV-2 N protein and chicken IgY which both labeled with latex microspheres. The test line contains the other monoclonal antibody against SARS-CoV-2 N protein. The control line contains rabbit-anti-chicken IgY antibody.
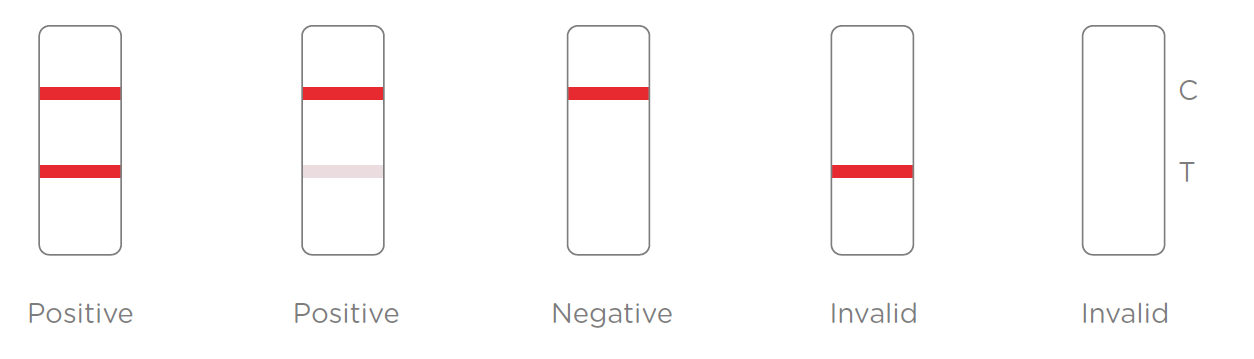
After the specimen is applied in the specimen well of the device, antigen in the specimen forms an immune complex with the binding reagent in the specimen zone. Then the complex migrates to the test zone. The test line in the test zone contains antibody from a specific pathogen. If the concentration of the specific antigen in the specimen is higher than LoD, it will be captured at the test line (T) and form a red line. In contrast, if the concentration of the specific antigen is lower than LoD, it will not form a red line. The test also contains an internal control system. A red control line (C) should always appear after the test is completed. Absence of a red control line indicates an invalid result.
Composition:
|
Composition |
Amount |
|
IFU |
1 |
|
Test cassette |
1/25 |
|
Extraction diluent |
1/25 |
|
Dropper tip |
1/25 |
|
Swab |
1/25 |
Test Procedure:
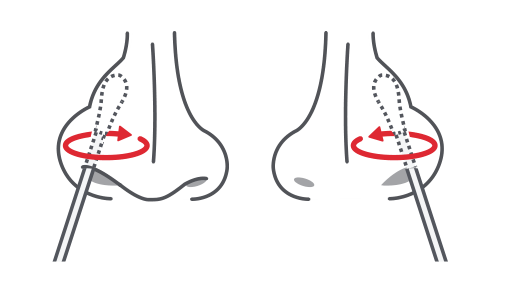
1.Specimen Collection
Take out the swab from the packaging without touching the padding. Carefully insert the cotton swab 1.5 cm into the nostril until slight resistance is noticeable. Do not insert the swab any deeper if you feel strong resistance or pain. Using moderate pressure, turn the swab 4 – 6 times in a circular motion for at least 15 seconds along the inner nasal wall in order to collect as many cells and mucus as possible. Repeat the sampling with the same swab in the other nostril.
2.Specimen Handling

3.Test Procedure

● Allow test device, specimen and diluent to equilibrate to room temperature 15~30℃ prior to opening the pouch. Remove the test device from the sealed aluminum foil pouch.
● Apply 3 drops of the test specimen into the specimen well.
● Wait for the red line(s) to appear at room temperature. Read results between 15~30 minutes. Do not read the result after 30 minutes.
Results Interpretation:
Product detail pictures:

Related Product Guide:
We have advanced equipment. Our products are exported to the USA, the UK and so on, enjoying a good reputation among customers for CE Certification Parainfluenza Virus Type 1 Manufacturers Exporters – 2019-nCoV Ag Test (Latex Chromatography Assay) / Professional Test / Anterior Nasal Swab – INNOVITA , The product will supply to all over the world, such as: Bogota, Lisbon, Tanzania, We pay high attention to customer service, and cherish every customer. We have maintained a strong reputation in the industry for many years. We are honest and work on building a long-term relationship with our customers.
We are old friends, the company's product quality has been always very good and this time the price is also very cheap.