-
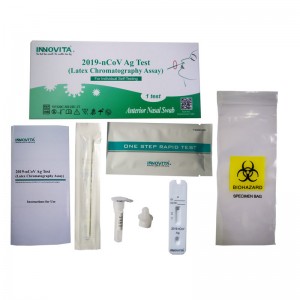
CE Certification China Rapid Antigen Swab Test Kit Manufacturers Exporters – 2019-nCoV Ag Test (Latex Chromatography Assay) / Self-Test / Anterior Nasal Swab – INNOVITA
Product Detail: Innovita® 2019-nCoV Ag Test is intended for the direct and qualitative detection of SARS-CoV-2 nucleocapsid protein antigen in anterior nasal swab that is self-collected by an individual aged 18 years or older or is collected by an adult from young individuals. It only recognizes the N protein and cannot detect the S protein or its mutation site. The kit is intended for layperson as self-testing at home or at work (in offices, for sports events, airports, schools, etc.) What ... -
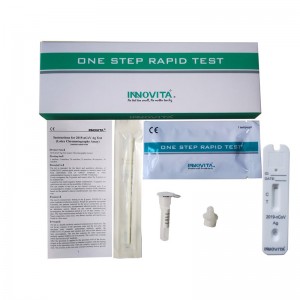
High-Quality OEM qualitative detection Factory Quotes – 2019-nCoV Ag Test (Latex Chromatography Assay) / Professional Test / Anterior Nasal Swab – INNOVITA
Product Detail: Innovita® 2019-nCoV Ag Test is intended for the direct and qualitative detection of SARS-CoV-2 nucleocapsid protein antigen in anterior nasal swabs from individuals who are suspected of COVID-19 by their healthcare provider within the first seven days of the onset of symptoms or for screening of individuals without symptoms or other reasons to suspect COVID-19 infection. The test results of this kit are for clinical reference only. It is recommended to conduct a comprehensive ... -
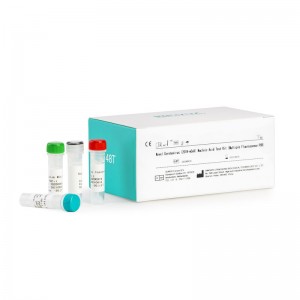
CE Certification Tropical Diseases Factories Pricelist – Novel Coronavirus (2019-nCoV) Nucleic Acid Test Kit (Multiple Fluorescence PCR) – INNOVITA
Product Detail: Innovita® 2019-nCoV IgM/IgG Test is intended for the diagnosis and epidemiological monitoring of diseases caused by the novel coronavirus (2019-nCoV). ORF1ab and N gene of 2019-nCoV are detected qualitatively from throat swabs and alveolar lavage fluid samples collected from suspected cases of pneumonia, patients with suspected cases, others who need to diagnose. The test results of this kit are for clinical reference only. It is recommended to conduct a comprehensive analysis... -
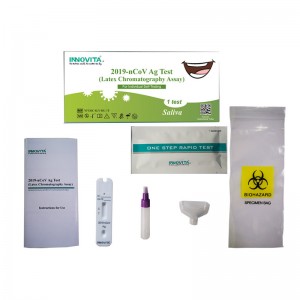
CE Certification Antigen Test Cassette Manufacturers Exporters – 2019-nCoV Ag Test (Latex Chromatography Assay) / Self-Test / Saliva – INNOVITA
Product Detail: Innovita® 2019-nCoV Ag Test is intended for the direct and qualitative detection of SARS-CoV-2 nucleocapsid protein antigen in saliva that is self-collected by an individual aged 18 years or older or is collected by an adult from young individuals. It only recognizes the N protein and cannot detect the S protein or its mutation site. The kit is intended for layperson as self-testing at home or at work (in offices, for sports events, airports, schools, etc.). What is self-test:... -
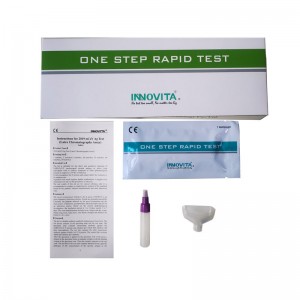
High-Quality OEM Immunofluorescence Assay Manufacturers Exporters – 2019-nCoV Ag Test (Latex Chromatography Assay) / Professional Test / Saliva – INNOVITA
Product Detail: Innovita® 2019-nCoV Ag Test is intended for the direct and qualitative detection of SARS-CoV-2 nucleocapsid protein antigen in saliva from individuals who are suspected of COVID-19 by their healthcare provider within the first seven days of the onset of symptoms or for screening of individuals without symptoms or other reasons to suspect COVID-19 infection. The test results of this kit are for clinical reference only. It is recommended to conduct a comprehensive analysis of th... -

CE Certification Neutralizing Antibody Manufacturers Exporters – 2019-nCoV Ag Test (Latex Chromatography Assay) / Professional Test / Saliva – INNOVITA
Product Detail: Innovita® 2019-nCoV Ag Test is intended for the direct and qualitative detection of SARS-CoV-2 nucleocapsid protein antigen in saliva from individuals who are suspected of COVID-19 by their healthcare provider within the first seven days of the onset of symptoms or for screening of individuals without symptoms or other reasons to suspect COVID-19 infection. The test results of this kit are for clinical reference only. It is recommended to conduct a comprehensive analysis of th... -

High-Quality OEM Saliva Test Manufacturers Exporters – 2019-nCoV Ag Test (Latex Chromatography Assay) / Professional Test / Saliva – INNOVITA
Product Detail: Innovita® 2019-nCoV Ag Test is intended for the direct and qualitative detection of SARS-CoV-2 nucleocapsid protein antigen in saliva from individuals who are suspected of COVID-19 by their healthcare provider within the first seven days of the onset of symptoms or for screening of individuals without symptoms or other reasons to suspect COVID-19 infection. The test results of this kit are for clinical reference only. It is recommended to conduct a comprehensive analysis of th... -

CE Certification Cardiac Marker Manufacturers Exporters – 2019-nCoV Ag Test (Latex Chromatography Assay) / Professional Test / Anterior Nasal Swab – INNOVITA
Product Detail: Innovita® 2019-nCoV Ag Test is intended for the direct and qualitative detection of SARS-CoV-2 nucleocapsid protein antigen in anterior nasal swabs from individuals who are suspected of COVID-19 by their healthcare provider within the first seven days of the onset of symptoms or for screening of individuals without symptoms or other reasons to suspect COVID-19 infection. The test results of this kit are for clinical reference only. It is recommended to conduct a comprehensive ... -

CE Certification Antigen Kit Factory Quotes – Novel Coronavirus (2019-nCoV) Nucleic Acid Test Kit (Multiple Fluorescence PCR) – INNOVITA
Product Detail: Innovita® 2019-nCoV IgM/IgG Test is intended for the diagnosis and epidemiological monitoring of diseases caused by the novel coronavirus (2019-nCoV). ORF1ab and N gene of 2019-nCoV are detected qualitatively from throat swabs and alveolar lavage fluid samples collected from suspected cases of pneumonia, patients with suspected cases, others who need to diagnose. The test results of this kit are for clinical reference only. It is recommended to conduct a comprehensive analysis... -

CE Certification Igm/Igg Factories Pricelist – Novel Coronavirus (2019-nCoV) Nucleic Acid Test Kit (Multiple Fluorescence PCR) – INNOVITA
Product Detail: Innovita® 2019-nCoV IgM/IgG Test is intended for the diagnosis and epidemiological monitoring of diseases caused by the novel coronavirus (2019-nCoV). ORF1ab and N gene of 2019-nCoV are detected qualitatively from throat swabs and alveolar lavage fluid samples collected from suspected cases of pneumonia, patients with suspected cases, others who need to diagnose. The test results of this kit are for clinical reference only. It is recommended to conduct a comprehensive analysis... -

CE Certification HPV Factory Quotes – 2019-nCoV Ag Test (Latex Chromatography Assay) / Self-Test / Saliva – INNOVITA
Product Detail: Innovita® 2019-nCoV Ag Test is intended for the direct and qualitative detection of SARS-CoV-2 nucleocapsid protein antigen in saliva that is self-collected by an individual aged 18 years or older or is collected by an adult from young individuals. It only recognizes the N protein and cannot detect the S protein or its mutation site. The kit is intended for layperson as self-testing at home or at work (in offices, for sports events, airports, schools, etc.). What is self-test:... -

High-Quality OEM Chlamydia Pneumonia Factory Quotes – Novel Coronavirus (2019-nCoV) Nucleic Acid Test Kit (Multiple Fluorescence PCR) – INNOVITA
Product Detail: Innovita® 2019-nCoV IgM/IgG Test is intended for the diagnosis and epidemiological monitoring of diseases caused by the novel coronavirus (2019-nCoV). ORF1ab and N gene of 2019-nCoV are detected qualitatively from throat swabs and alveolar lavage fluid samples collected from suspected cases of pneumonia, patients with suspected cases, others who need to diagnose. The test results of this kit are for clinical reference only. It is recommended to conduct a comprehensive analysis...






